Benefits of vaccination
Vaccines have saved millions of lives in Europe and across the world. Widespread vaccination has helped to eliminate smallpox and polio from Europe and made previously common diseases become rare.
By getting vaccinated, people are protected against serious illness, and sometimes death, from vaccine-preventable diseases. It also protects against spreading those diseases among family members, friends, colleagues, classmates and the wider community.
Vaccination is considered one of the most cost-effective public health interventions, as it prevents the costs associated with treating and caring for people who become ill.
When people in a population are vaccinated against an infectious disease, it becomes much less likely to spread from person to person. This protects against outbreaks of disease and, when enough people become immune, can enable ‘community immunity’ or ‘herd immunity’. This helps to protect not only those vaccinated but also, indirectly, those who are more vulnerable to disease. Young children, the elderly, people with weak immune systems, cancer patients, and people who cannot be vaccinated for medical reasons are all at risk when diseases start spreading through communities, so getting vaccinated can help protect them as well.
How contagious are different diseases?
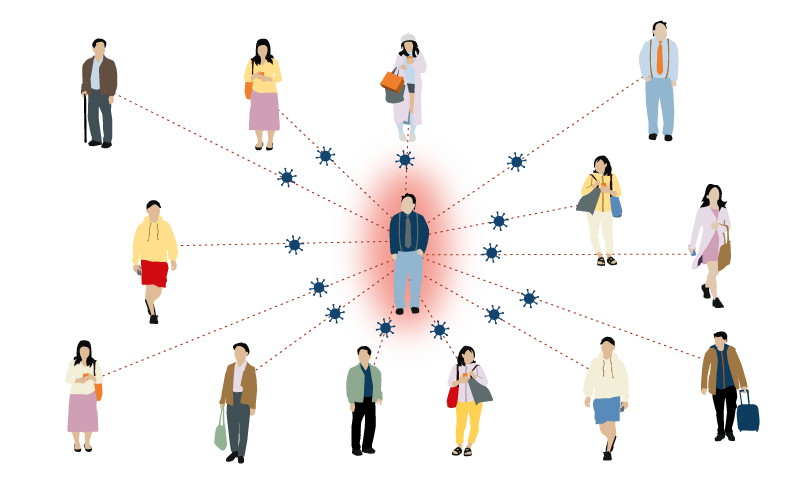
Measles
12-18 people
One person with measles can infect between 12-18 people.
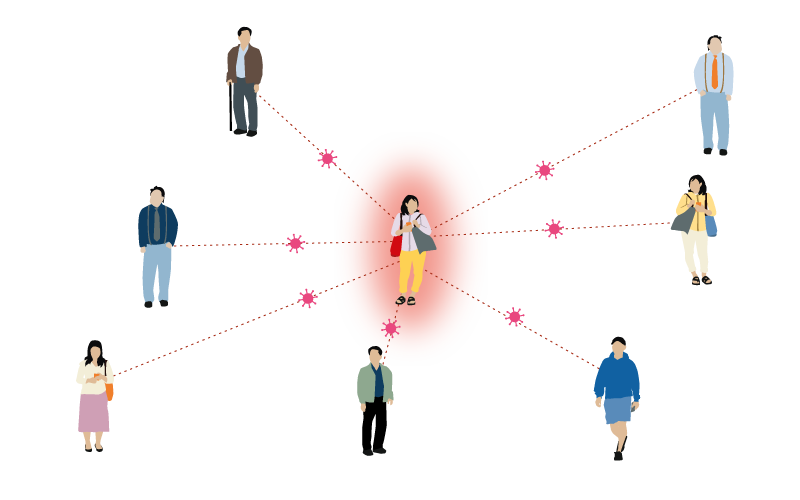
Mumps
4-7 people
One person with mumps can infect between 4-7 people.
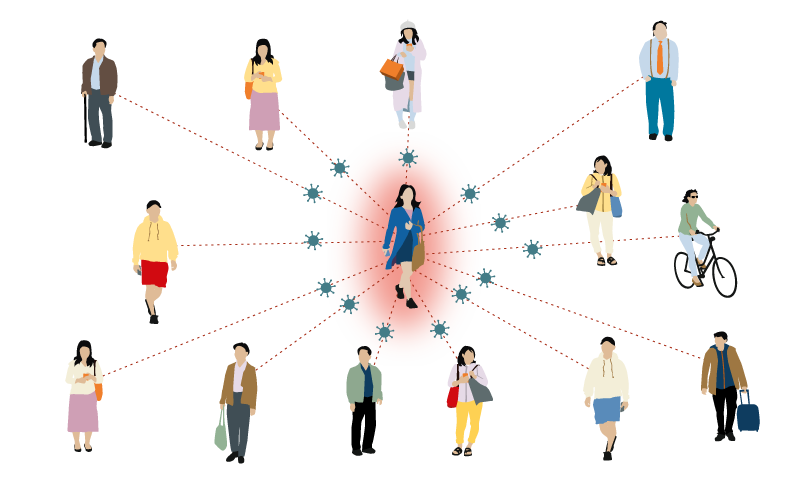
Whooping cough
12-17 people
One person with whooping cough can infect between 12-17 people.
Diphtheria/Rubella
6-7 people
One person with diphtheria or rubella can infect between 6-7 people.

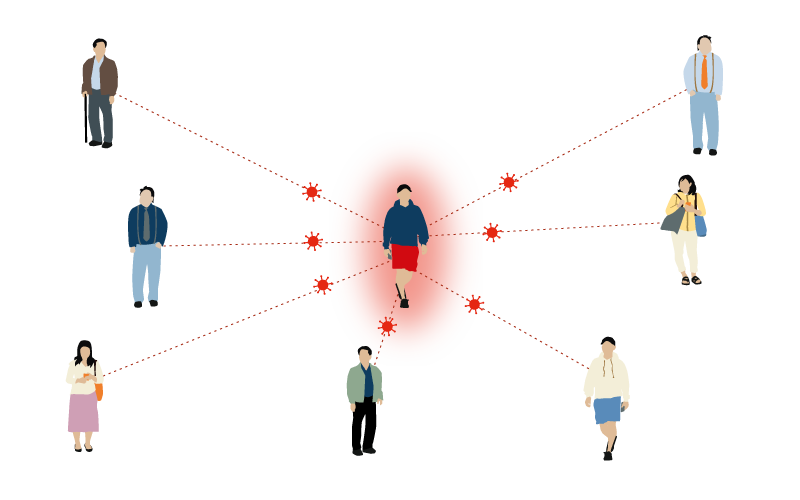
Influenza
1-4 people
One person with influenza can infect between 1-4 people.
For some infectious diseases, people cannot rely on community immunity. Tetanus is one such example, as it can develop as a result of common injuries such as cuts, scrapes and dog or cat bites. Vaccination is the only way to ensure direct protection against tetanus.
Unlike a treatment given to cure an illness, vaccines are usually given to healthy people to prevent them from getting ill. Although the long-term benefits of getting vaccinated may not be immediately obvious, if people stopped being vaccinated, many of the diseases that have become very rare today thanks to vaccination, could return.
